How Many Neutrons Are in an Atom of Mg 25
All isotopes of Magnesium have 12 protons therefore Mg-25 has 25 - 12 13 neutrons in its nucleus. How many neutrons are in an atom of mg 25 13 neutronsMass25Number12Mass - Number Amount of NeutronsThe atomic number is also the mount of Protons and Electrons of the element.

This Is The Magnesium 24 Isotope It Has 12 Neutrons And 12 Protons The Other Two Most Popular Isotopes Ar Atom Project Element Symbols Electron Configuration
One atom of Mg-25 has 25 nucleons hence the 25 in its name.

. How many protons neutrons and electrons are in mg with an atomic mass number of 25. 17 electrons What is the cost of sodium. If mass number number of protons number of neutrons then number of neutrons mass number - number of protons or atomic number.
Atomic number means 12 protons. For Magnesium atomic number 12 Number of neutrons 25 - 12 13 neutrons. For a negatively charged ion add the negative charge value with the number of electrons of the neutral atom.
D a Calcium Ca2 into the mix. Zn-64 Atomic Number Z 30 Mass Number A 64 Nucleon Number A 64 Proton Number Z 30. There are 12 protons and 13 neutrons in an atom of magnesium-25.
How many protons does Zinc-64. So 25 - 12 13 neutrons. How many neutrons are in an atom of Mg-25.
1500 grams of asf3 were reacted with 1800 g of ccl4 to produce ascl3 and ccl2f2. We can determine this by subtracting the number of protons in the atom from the atomic mass which How many electrons does chlorine 37. Imagine you mix together a few different pure elements.
If the actual yield of ccl2f2 was 127 g what is the percent yield. 30 Properties of Zn-64 Isotope. Mass of Mg is 25 amu.
There are 13 neutrons in an atom of magnesium-25. 25 is the number of protons and neutrons added together. A-NZ 25-1312 The atomic number of the element is 12.
The atomic number of Mg is 12 which is the number of protons. If you need 05L of 01M Cuso4 to perform another reaction explain how you would prepare. The mass number of an isotope is the sum of the protons and neutrons in its atomic nuclei.
Mass number of an element is equal to the sum of number of protons and number of neutrons Hence mass number 5 7 12. New questions in Chemistry. Thus there are 13 neutron in Mg atom.
So the difference of 25-12 13. 7 rows Mass numbers of typical isotopes of Magnesium are 24. 2 Show answers Another question on Chemistry.
Atomic number of Mg is 12. How many neutrons are in an atom of mg-25 - 1726571 Question 5 1 point A low pressure tropical storm that forms over warm ocean water causing a strong whirling storm is a. How many neutrons are in hydrogen.
For boron-11 this total is 11 and five of the particles are protons thus 1156 neutrons. So boron-11 has five protons the same as boron-10. How many neutrons are in an atom of mg-25.
If we know the mass number A and the number of neutrons N we can calculate the atomic number Z by subtracting the number of neutrons from the mass number. K which forms a 1 ion Mg which forms a 2 ion Cl which forms a-1 ion Now you ad. Then the mass number is total protons plus neutrons.
How many neutrons are in an atom of Mg-25. The total number of neutrons in.
How Many Neutrons In Magnesium Quora
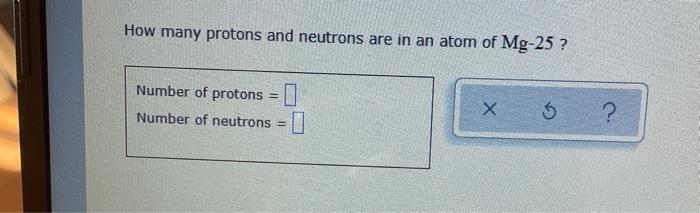
Solved How Many Protons And Neutrons Are In An Atom Of Mg 25 Chegg Com
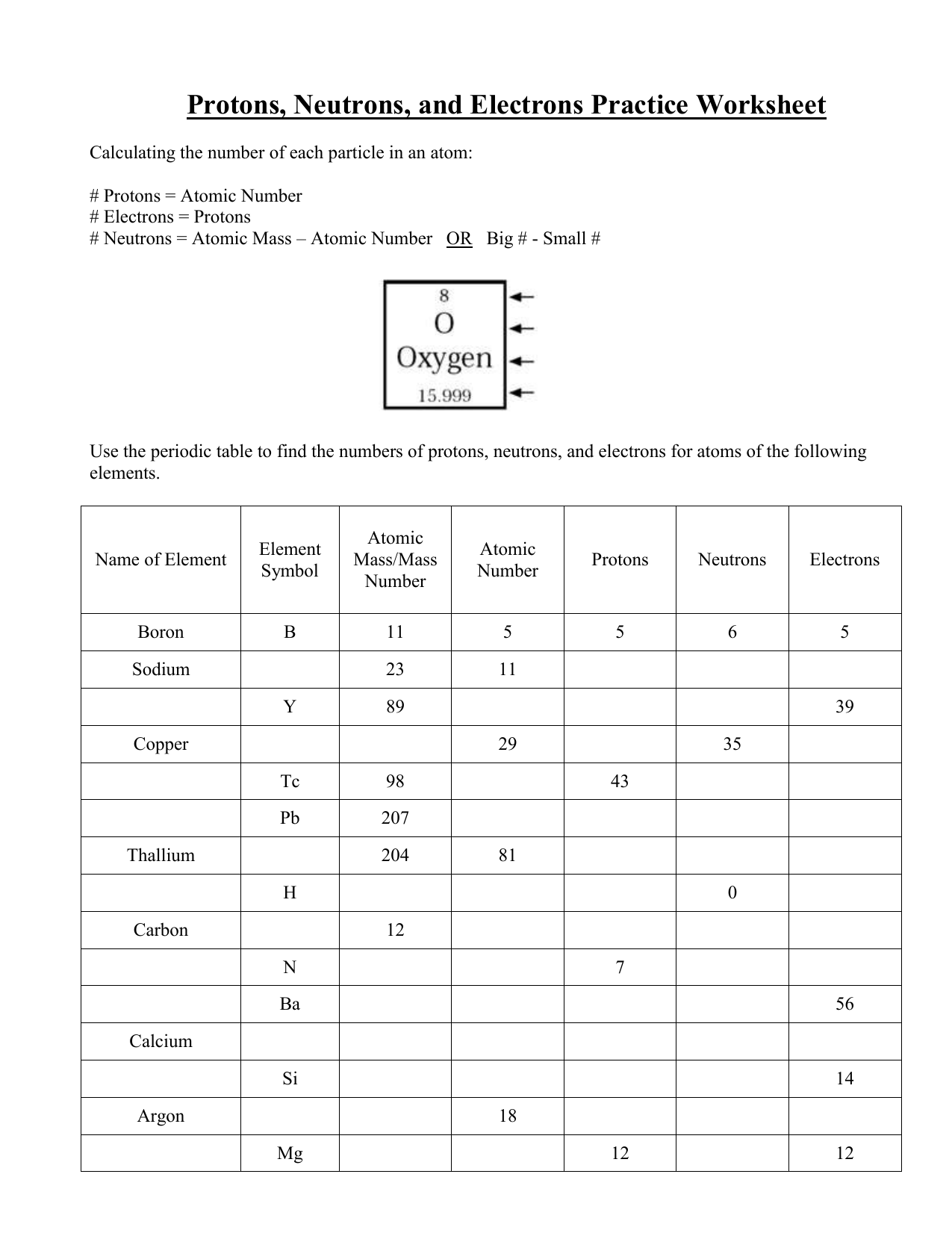
Protons Neutrons And Electrons
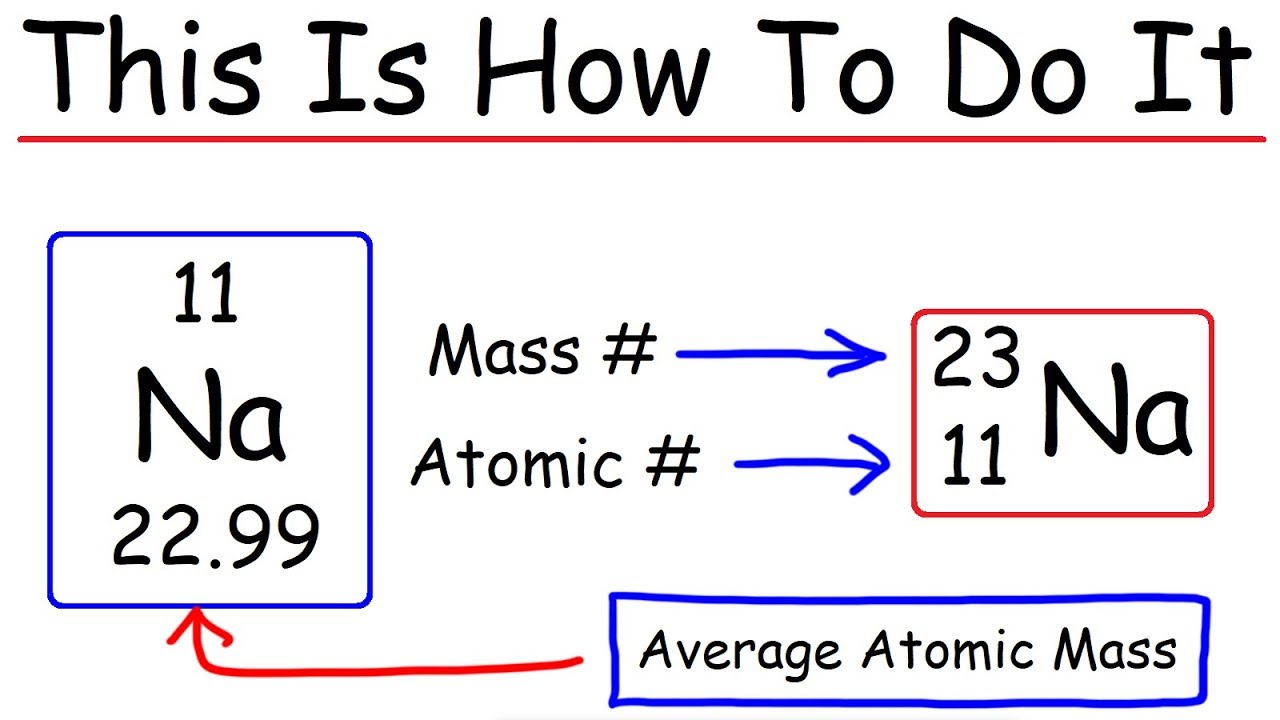
How To Calculate The Number Of Protons Neutrons And Electrons Chemistry Youtube
No comments for "How Many Neutrons Are in an Atom of Mg 25"
Post a Comment